economy /
Which catalyst is used in cracking process?
Modern cracking uses zeolites as the catalyst. These are complex aluminosilicates, and are large lattices of aluminium, silicon and oxygen atoms carrying a negative charge. They are, of course, associated with positive ions such as sodium ions.
What is used as a good catalyst in petroleum cracking?
In sulfuric acid alkylation, concentrated sulfuric acid of 98 percent purity serves as the catalyst for a reaction that is carried out at 2 to 7 °C (35 to 45 °F).Which type of catalyst is used in catalytic cracking Mcq?
Which type of catalyst is used in catalytic cracking? Explanation: Earlier acid clays were used as a catalyst for catalytic cracking. Now silica and alumina with minor amounts of oxides of Ca, Mg, Na and rare earth are used as a catalyst.Which of the following is used as a catalyst in fluidized bed catalytic cracking?
A modern FCC catalyst has four major components: crystalline zeolite, matrix, binder, and filler. Zeolite is the active component and can comprise from about 15 to 50 weight percent of the catalyst. Faujasite (aka Type Y) is the zeolite used in FCC units.What is the process of cracking?
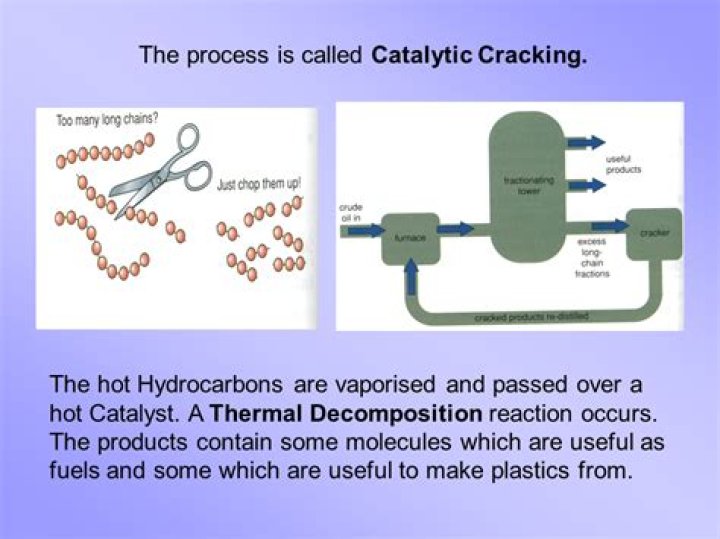
Cracking is a reaction in which larger saturated hydrocarbon molecules are broken down into smaller, more useful hydrocarbon molecules, some of which are unsaturated: the original starting hydrocarbons are alkanes. the products of cracking include alkanes and alkenes , members of a different homologous series.Cracking of Petroleum (Catalytic Cracking)
Why are catalysts used in cracking?
As the name implies, the catalytic cracking process takes place in the presence of a catalyst. The catalyst promotes breaking of the large molecules in a certain way that maximizes the yield of gasoline. Ideally, all of the “cat-cracked” product would be in the gasoline range, but the technology is not that capable.Why are catalysts used in catalytic cracking?
5.5.In catalytic cracking solid catalysts effectively convert plastics into liquid fuel, giving lighter fractions as compared to thermal cracking. Catalysts used in this process include silica–alumina, aluminosilicate zeolite (ZSM-5), and fluid catalytic cracking (FCC), among others.
Why are zeolites used in cracking?
Catalysts called zeolites drive cracking. These mineral-like solids of silicon, aluminium and oxygen contain orderly networks of pores, just wide enough for the hydrocarbon molecules to wriggle down. Inside a zeolite's pores, it's extremely acidic. This breaks up the big molecules.Does thermal cracking use a catalyst?
These use completely different sets of conditions. Thermal cracking does not go via ionic intermediates like catalytic cracking. Instead, carbon-carbon bonds are broken so that each carbon atom ends up with a single electron. In other words, free radicals are formed.How many catalysts are there?
They are (1) Homogeneous, (2) Heterogeneous (solid), (3) Heterogenized homogeneous catalyst and (4) Biocatalysts.What is catalytic cracking in organic chemistry?
Catalytic cracking is an important process in the oil industry where petroleum vapor passes through a low-density bed of catalyst, which causes the heavier fractions to 'crack' producing lighter more valuable products. In the petrochemicals industry they are used for producing polyolefins on a very large scale.Why is aluminium oxide used in cracking?
The larger alkanes are heated to around 650°C and their vapours are passed over a hot catalyst containing aluminium oxide. This causes covalent bonds to break and reform.Why is porcelain used in cracking?
Caption. Cracking liquid paraffin. In the horizontal test tube are a piece of mineral wool (brown) soaked in liquid paraffin, and porcelain chips, which act as a catalyst. As the porcelain chips are heated the vapour from the paraffin is 'cracked', or broken down into smaller hydrocarbons.What type of reaction is cracking?
Cracking is an example of a thermal decomposition chemical reaction.What are the 3 types of catalyst?
Catalysts can be categorized as homogeneous, heterogeneous, or enzymatic. Homogeneous catalysts exist in the same phase as the reactants, whereas heterogeneous catalysts exist in a different phase than the reactants.What is the most common catalyst?
Enzymes are naturally occurring catalysts responsible for many essential biochemical reactions. Most solid catalysts are metals or the oxides, sulfides, and halides of metallic elements and of the semimetallic elements boron, aluminum, and silicon.What are common catalysts?
Common types of catalysts include enzymes, acid-base catalysts, and heterogeneous (or surface) catalysts.What are examples of catalyst?
Examples of Inorganic Catalysts
- palladium - If you light a match in a room with hydrogen gas and oxygen gas, there will be an explosion and most of the hydrogen and oxygen will combine to create water molecules.
- iron - When making ammonia, iron is a helpful catalyst.